Clinical Trials
Success Measurements
Strategic Initiative
Clinical trials are essential for the study of new methods of cancer prevention, detection and diagnosis, and treatment. While cancer drug trials are often most publicized, clinical trials have few limits in their scope and can be used to assess many different types of interventions, for example:
• The impact of different surgical practices
• The role of physical therapy
• The role of cognitive therapy
• The impact of supportive services (counselling, nurse specialists)
• Strategies to improve treatment tolerance and reduce side effects
• The impact of new predictive and prognostic factors on patient outcomes
• The value of new biomarkers in patient treatment
• The impact of changes in practices and processes in treating gynecologic cancers
The planning and conduct of contemporary clinical trials must include patients as partners and not just as subjects. It is expected that patients will now participate in the design, conduct and evaluation of successful clinical trials. This past year, the GCI established a Gynecologic Cancer Patient & Family Advisory Council to ensure that the patient voice is a part of all studies.
Clinical trials engaging women affected by or at risk for gynecologic cancer in BC are done across multiple sites including the largest at BC Cancer and at BC Women’s Hospital + Health Centre, the Vancouver General Hospital and in cooperation with the UBC Faculty of Medicine.
There is also the need to develop capacity to conduct trials in the regional centres across the Province including Abbotsford, Kelowna, Prince George, Surrey, Vancouver and Victoria. A strong clinical trials program is fundamental in cancer research and care. Clinical trials are the essential final step for testing important scientific advances and for improving the standard of care for patients.
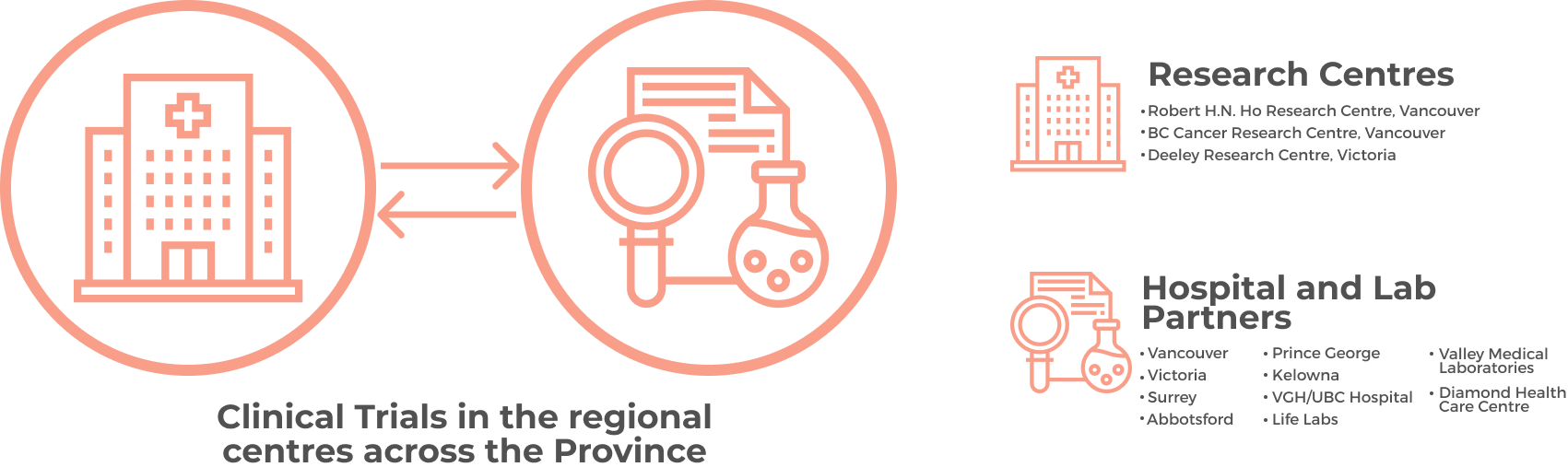
The GCI Clinical Trials Group will serve as a hub from which innovative new ideas and studies will be carried out throughout BC by clinicians and researchers in the laboratory and in partnership with the patients. To ensure equitable access to clinical studies and to promote collaboration across the province, the trials and studies may be pan-provincial, across multiple health authorities and institutions, diverse in the populations to be studied and ethical in their execution. Given the substantial strength in molecular biology and translational research in gynecologic cancers amongst our group, priority will be given to trials that leverage this basic understanding to improve patient outcomes, including:
• Translational studies with strong emphasis on personalized patient care
• Drug development and testing of novel therapies
• Screening and prevention trials
• Survivorship studies
• Assessment of imaging modalities
• Assessment of radiation therapy techniques
• Patient oriented research surveys
• Patient reported outcome measures

I. Strengthening the Environment for Clinical Trials in BC
To advance the key priority of clinical research, the GCI sees the recruitment of additional talent to spearhead new gynecologic cancer trials as the next crucial step that must be taken to build a comprehensive gynecological cancer research program and achieve global impact to improve outcomes for women with gynecologic cancers. The additional people resources will further develop a coordinated, fully collaborative clinical research agenda and create new opportunities in collaboration with the clinical and research team.
Technology driven research will play a key role in the GCI’s success. There is an urgent need for a mass spectrometer and the operating costs and talent to sustain it. The current shared use of this precision equipment for clinical trials requires a lead time of over a year, which is precious time to a newly-diagnosed patient. The dedicated equipment will increase capacity and will enable its use as an adjunct for enrolling gynecologic cancer patients in clinical trials. Once proven in clinical testing, this approach can then be tested prospectively as a decision tool to guide patient care.
Clinical trials are an expensive process as they require protocol development and review, Health Canada approval, contracting between organizations, access to Medidata Rave (an FDA compliant, state-of-the art, web-based electronic data capture system), trial coordination, auditing and data input and reporting (e.g. toxicity), data safety monitoring (e.g. severe adverse events), centralized biobanking, expert pathology review, diagnostic assays and imaging, and any additional tests (e.g. cardiac scan) and biopsies for patients enrolled in the gynecologic clinical trials. For this reason, it is important that the GCI works closely with the resources through Clinical Trials BC in order to optimize our environment for high quality trials (https://www.clinicaltrialsbc.ca/).
BC has a history of successful clinical trials in gynecologic cancers. This advanced program schedule for future trials will only solidify BC’s position as a centre of excellence in gynecologic cancer research worldwide.
II. Personalized Care and Treatment
Treatment of gynecologic cancers is still largely generic without consideration of specific molecular features that contribute to disease progression and treatment response. The co-development of biomarkers and treatment research will be fostered through the GCI to ensure that the development and validation of predictive markers are carried through to the clinical setting by enabling access to clinical trials. We believe that translation of precision therapeutics requires tight integration between biomarker development and classification and clinical trials. This is a critical moment for the research team in BC as their genomic research requires further study in patients to see if the important findings can influence treatment selection and patient outcomes.
III. Establishing a Rare Gynecological Cancers Program
As 40% of women’s reproductive cancer types are rare (i.e. incidence of <15/100,000/year, as defined by the National Cancer Institute), one of the strategic priorities will be to develop pan-Canadian Rare Reproductive Cancers Program. This Program will support Canadian research in rare reproductive cancers to expedite translation. With leading Genomics Centre expertise located in Vancouver, Toronto and Montreal, the GCI will facilitate genomic profiling for women enrolled in this program, allowing molecular features to be considered in selection of treatments. The pan-Canadian reach of this program will maximize enrollment into rare cancer trials, an important consideration as no single centre has enough cases.
Molecular features and diagnostic information will be used to connect patients to existing clinical trials being conducted across BC and Canada and development of new trial opportunities, including basket trials to group multiple tumour types with similar molecular features to compensate for the small numbers of women affected by these cancers.
